what organelle captures energy from the sun to make food?
All animals and most microorganisms rely on the continual uptake of large amounts of organic compounds from their environment. These compounds are used to provide both the carbon skeletons for biosynthesis and the metabolic energy that drives cellular processes. Information technology is believed that the get-go organisms on the archaic Earth had access to an abundance of the organic compounds produced by geochemical processes, only that most of these original compounds were used upwards billions of years ago. Since that fourth dimension, the vast majority of the organic materials required by living cells have been produced by photosynthetic organisms, including many types of photosynthetic bacteria.
The most advanced photosynthetic bacteria are the cyanobacteria, which have minimal nutrient requirements. They use electrons from water and the energy of sunlight when they convert atmospheric COii into organic compounds—a process called carbon fixation. In the course of splitting h2o [in the overall reaction northH2O + northCO2

(CHiiO) n + nOii], they also liberate into the temper the oxygen required for oxidative phosphorylation. As we meet in this section, information technology is thought that the evolution of cyanobacteria from more than primitive photosynthetic bacteria eventually fabricated possible the development of abundant aerobic life forms.
In plants and algae, which adult much later, photosynthesis occurs in a specialized intracellular organelle—the chloroplast. Chloroplasts perform photosynthesis during the daylight hours. The immediate products of photosynthesis, NADPH and ATP, are used by the photosynthetic cells to produce many organic molecules. In plants, the products include a low-molecular-weight saccharide (commonly sucrose) that is exported to encounter the metabolic needs of the many nonphotosynthetic cells of the organism.
Biochemical and genetic evidence strongly propose that chloroplasts are descendants of oxygen-producing photosynthetic bacteria that were endocytosed and lived in symbiosis with primitive eucaryotic cells. Mitochondria are besides generally believed to be descended from an endocytosed bacterium. The many differences betwixt chloroplasts and mitochondria are idea to reflect their different bacterial ancestors, as well every bit their subsequent evolutionary divergence. Nevertheless, the fundamental mechanisms involved in light-driven ATP synthesis in chloroplasts are very similar to those that we accept already discussed for respiration-driven ATP synthesis in mitochondria.
The Chloroplast Is I Member of the Plastid Family of Organelles
Chloroplasts are the about prominent members of the plastid family unit of organelles. Plastids are present in all living found cells, each cell blazon having its own feature complement. All plastids share certain features. Most notably, all plastids in a particular constitute species contain multiple copies of the same relatively small genome. In add-on, each is enclosed by an envelope composed of two concentric membranes.
As discussed in Chapter 12 (see Figure 12-3), all plastids develop from proplastids, small organelles in the young cells of found meristems (Figure 14-33A). Proplastids develop according to the requirements of each differentiated cell, and the type that is nowadays is determined in large part by the nuclear genome. If a foliage is grown in darkness, its proplastids enlarge and develop into etioplasts, which accept a semicrystalline array of internal membranes containing a yellow chlorophyll precursor instead of chlorophyll. When exposed to light, the etioplasts rapidly develop into chloroplasts past converting this precursor to chlorophyll and by synthesizing new membrane pigments, photosynthetic enzymes, and components of the electron-send chain.

Effigy 14-33
Plastid diversity. (A) A proplastid from a root tip jail cell of a edible bean plant. Notation the double membrane; the inner membrane has also generated the relatively sparse internal membranes present. (B) Iii amyloplasts (a grade of leucoplast), or starch-storing (more than...)
Leucoplasts are plastids present in many epidermal and internal tissues that exercise not become green and photosynthetic. They are picayune more than than enlarged proplastids. A common form of leucoplast is the amyloplast (Figure 14-33B), which accumulates the polysaccharide starch in storage tissues—a source of sugar for future use. In some plants, such as potatoes, the amyloplasts can grow to be as large every bit an average animal cell.
It is important to realize that plastids are not just sites for photosynthesis and the deposition of storage materials. Plants have also used their plastids to compartmentalize their intermediary metabolism. Purine and pyrimidine synthesis, most amino acid synthesis, and all of the fatty acid synthesis of plants takes place in the plastids, whereas in animal cells these compounds are produced in the cytosol.
Chloroplasts Resemble Mitochondria But Accept an Extra Compartment
Chloroplasts bear out their energy interconversions by chemiosmotic mechanisms in much the same way that mitochondria do. Although much larger (Effigy 14-34A), they are organized on the aforementioned principles. They have a highly permeable outer membrane; a much less permeable inner membrane, in which membrane ship proteins are embedded; and a narrow intermembrane space in between. Together, these membranes grade the chloroplast envelope (Effigy fourteen-34B,C). The inner membrane surrounds a large space called the stroma, which is coordinating to the mitochondrial matrix and contains many metabolic enzymes. Like the mitochondrion, the chloroplast has its own genome and genetic system. The stroma therefore as well contains a special gear up of ribosomes, RNAs, and the chloroplast Dna.
Figure xiv-34
Electron micrographs of chloroplasts. (A) In a wheat foliage jail cell, a sparse rim of cytoplasm—containing chloroplasts, the nucleus, and mitochondria—surrounds a big vacuole. (B) A thin section of a single chloroplast, showing the chloroplast (more...)
In that location is, however, an important difference between the system of mitochondria and that of chloroplasts. The inner membrane of the chloroplast is not folded into cristae and does non contain electron-send chains. Instead, the electron-transport bondage, photosynthetic lite-capturing systems, and ATP synthase are all contained in the thylakoid membrane, a third distinct membrane that forms a set of flattened disclike sacs, the thylakoids (Figure 14-35). The lumen of each thylakoid is thought to be connected with the lumen of other thylakoids, thereby defining a third internal compartment called the thylakoid space, which is separated by the thylakoid membrane from the stroma that surrounds it.

Figure fourteen-35
The chloroplast. This photosynthetic organelle contains three singled-out membranes (the outer membrane, the inner membrane, and the thylakoid membrane) that ascertain three split internal compartments (the intermembrane space, the stroma, and the thylakoid (more...)
The structural similarities and differences between mitochondria and chloroplasts are illustrated in Figure 14-36. The head of the chloroplast ATP synthase, where ATP is made, protrudes from the thylakoid membrane into the stroma, whereas it protrudes into the matrix from the inner mitochondrial membrane.

Figure xiv-36
A mitochondrion and chloroplast compared. A chloroplast is generally much larger than a mitochondrion and contains, in addition to an outer and inner membrane, a thylakoid membrane enclosing a thylakoid infinite. Unlike the chloroplast inner membrane, the (more than...)
Chloroplasts Capture Free energy from Sunlight and Use Information technology to Set Carbon
The many reactions that occur during photosynthesis in plants tin be grouped into two broad categories:
- 1.
-
In the photosynthetic electron-transfer reactions (also called the "lite reactions"), free energy derived from sunlight energizes an electron in the green organic paint chlorophyll, enabling the electron to move along an electron-transport chain in the thylakoid membrane in much the same manner that an electron moves along the respiratory chain in mitochondria. The chlorophyll obtains its electrons from water (H2O), producing O2 as a by-product. During the electron-transport process, H+ is pumped across the thylakoid membrane, and the resulting electrochemical proton gradient drives the synthesis of ATP in the stroma. As the terminal step in this series of reactions, high-energy electrons are loaded (together with H+) onto NADP+, converting it to NADPH. All of these reactions are confined to the chloroplast.
- 2.
-
In the carbon-fixation reactions (besides called the "dark reactions"), the ATP and the NADPH produced by the photosynthetic electron-transfer reactions serve as the source of free energy and reducing power, respectively, to drive the conversion of COii to carbohydrate. The carbon-fixation reactions, which brainstorm in the chloroplast stroma and continue in the cytosol, produce sucrose and many other organic molecules in the leaves of the institute. The sucrose is exported to other tissues equally a source of both organic molecules and energy for growth.
Thus, the germination of ATP, NADPH, and O2 (which requires light energy directly) and the conversion of CO2 to carbohydrate (which requires calorie-free energy merely indirectly) are separate processes (Figure 14-37), although elaborate feedback mechanisms interconnect the two. Several of the chloroplast enzymes required for carbon fixation, for example, are inactivated in the dark and reactivated past lite-stimulated electron-transport processes.
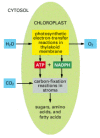
Figure 14-37
The reactions of photosynthesis in a chloroplast. Water is oxidized and oxygen is released in the photosynthetic electron-transfer reactions, while carbon dioxide is assimilated (stock-still) to produce sugars and a variety of other organic molecules in the (more...)
Carbon Fixation Is Catalyzed by Ribulose Bisphosphate Carboxylase
Nosotros take seen before in this affiliate how cells produce ATP by using the large corporeality of free energy released when carbohydrates are oxidized to COtwo and H2O. Clearly, therefore, the opposite reaction, in which COtwo and H2O combine to make carbohydrate, must be a very unfavorable ane that can simply occur if it is coupled to other, very favorable reactions that drive information technology.
The central reaction of carbon fixation, in which an cantlet of inorganic carbon is converted to organic carbon, is illustrated in Figure 14-38: COtwo from the atmosphere combines with the five-carbon compound ribulose 1,v-bisphosphate plus water to yield ii molecules of the three-carbon compound 3-phosphoglycerate. This "carbon-fixing" reaction, which was discovered in 1948, is catalyzed in the chloroplast stroma by a large enzyme called ribulose bisphosphate carboxylase. Since each molecule of the circuitous works sluggishly (processing just about three molecules of substrate per second compared to yard molecules per second for a typical enzyme), many enzyme molecules are needed. Ribulose bisphosphate carboxylase oft constitutes more fifty% of the full chloroplast protein, and information technology is idea to be the most abundant protein on Earth.
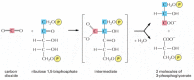
Effigy 14-38
The initial reaction in carbon fixation. This reaction, in which carbon dioxide is converted into organic carbon, is catalyzed in the chloroplast stroma by the arable enzyme ribulose bisphosphate carboxylase. The product is 3-phosphoglycerate, which (more...)
Three Molecules of ATP and Two Molecules of NADPH Are Consumed for Each CO2 Molecule That Is Stock-still
The actual reaction in which CO2 is stock-still is energetically favorable because of the reactivity of the energy-rich compound ribulose 1,5-bisphosphate, to which each molecule of COtwo is added (see Effigy 14-38). The elaborate metabolic pathway that produces ribulose ane,5-bisphosphate requires both NADPH and ATP; it was worked out in one of the first successful applications of radioisotopes as tracers in biochemistry. This carbon-fixation cycle (also called the Calvin cycle) is outlined in Figure 14-39. It starts when 3 molecules of CO2 are stock-still past ribulose bisphosphate carboxylase to produce half-dozen molecules of 3-phosphoglycerate (containing half-dozen × 3 = eighteen carbon atoms in all: three from the COtwo and 15 from ribulose one,5-bisphosphate). The 18 carbon atoms then undergo a cycle of reactions that regenerates the 3 molecules of ribulose ane,5-bisphosphate used in the initial carbon-fixation step (containing 3 × 5 = 15 carbon atoms). This leaves i molecule of glyceraldehyde 3-phosphate (3 carbon atoms) equally the net gain.
Figure 14-39
The carbon-fixation cycle, which forms organic molecules from COtwo and H2O. The number of carbon atoms in each type of molecule is indicated in the white box. There are many intermediates betwixt glyceraldehyde 3-phosphate and ribulose five-phosphate, simply (more than...)
A total of three molecules of ATP and two molecules of NADPH are consumed for each CO2 molecule converted into carbohydrate. The net equation is:

Thus, both phosphate-bond energy (as ATP) and reducing power (as NADPH) are required for the germination of organic molecules from COii and H2O. We return to this important betoken later.
The glyceraldehyde 3-phosphate produced in chloroplasts by the carbon-fixation bike is a 3-carbon sugar that besides serves as a central intermediate in glycolysis. Much of it is exported to the cytosol, where information technology can be converted into fructose vi-phosphate and glucose 1-phosphate by the reversal of several reactions in glycolysis (encounter Panel 2-8, pp. 124–125). The glucose one-phosphate is then converted to the sugar nucleotide UDP-glucose, and this combines with the fructose 6-phosphate to form sucrose phosphate, the immediate forerunner of the disaccharide sucrose. Sucrose is the major form in which sugar is transported between plant cells: just as glucose is transported in the claret of animals, sucrose is exported from the leaves via vascular bundles, providing the saccharide required by the balance of the establish.
Near of the glyceraldehyde three-phosphate that remains in the chloroplast is converted to starch in the stroma. Similar glycogen in animal cells, starch is a large polymer of glucose that serves as a carbohydrate reserve (run across Figure 14-33B). The product of starch is regulated and so that it is produced and stored as big grains in the chloroplast stroma during periods of excess photosynthetic chapters. This occurs through reactions in the stroma that are the reverse of those in glycolysis: they convert glyceraldehyde 3-phosphate to glucose 1-phosphate, which is and so used to produce the carbohydrate nucleotide ADP-glucose, the immediate precursor of starch. At night the starch is cleaved down to help support the metabolic needs of the plant. Starch provides an important part of the diet of all animals that eat plants.
Carbon Fixation in Some Plants Is Compartmentalized to Facilitate Growth at Low CO2 Concentrations
Although ribulose bisphosphate carboxylase preferentially adds CO2 to ribulose 1,5-bisphosphate, it can use Oii as a substrate in place of CO2, and if the concentration of CO2 is low, it will add together Otwo to ribulose ane,5-bisphosphate instead (see Figure 14-38). This is the first step in a pathway called photorespiration, whose ultimate issue is to use up O2 and liberate CO2 without the production of useful free energy stores. In many plants, about 1-third of the CO2 fixed is lost once again as COtwo considering of photorespiration.
Photorespiration tin exist a serious liability for plants in hot, dry weather condition, which cause them to shut their stomata (the gas exchange pores in their leaves) to avoid excessive water loss. This in plow causes the CO2 levels in the leaf to fall precipitously, thereby favoring photorespiration. A special accommodation, nevertheless, occurs in the leaves of many plants, such as corn and sugar cane that alive in hot, dry environments. In these plants, the carbon-fixation cycle occurs simply in the chloroplasts of specialized bundle-sheath cells, which contain all of the institute's ribulose bisphosphate carboxylase. These cells are protected from the air and are surrounded by a specialized layer of mesophyll cells that use the free energy harvested by their chloroplasts to "pump" COii into the bundle-sheath cells. This supplies the ribulose bisphosphate carboxylase with a high concentration of COii, thereby greatly reducing photorespiration.
The CO2 pump is produced by a reaction wheel that begins in the cytosol of the mesophyll cells. A CO2-fixation step is catalyzed past an enzyme that binds carbon dioxide (every bit bicarbonate) and combines it with an activated iii-carbon molecule to produce a iv-carbon molecule. The iv-carbon molecule diffuses into the bundle-sheath cells, where information technology is broken down to release the CO2 and generate a molecule with iii carbons. The pumping cycle is completed when this three-carbon molecule is returned to the mesophyll cells and converted back to its original activated form. Considering the CO2 is initially captured past converting it into a compound containing four carbons, the COtwo-pumping plants are called Civ plants. All other plants are called C3 plants because they capture CO2 into the 3-carbon chemical compound 3-phosphoglycerate (Figure xiv-40).

Effigy fourteen-twoscore
Comparative leaf anatomy in a Cthree plant and a Civ plant. The cells with green cytosol in the leaf interior contain chloroplasts that perform the normal carbon-fixation cycle. In C4 plants, the mesophyll cells are specialized for CO2 pumping rather than (more than...)
As for whatsoever vectorial transport procedure, pumping COtwo into the bundle-sheath cells in C4 plants costs energy. In hot, dry environments, all the same, this toll can be much less than the free energy lost by photorespiration in C3 plants, so C4 plants have a potential advantage. Moreover, because Cfour plants can perform photosynthesis at a lower concentration of CO2 inside the foliage, they need to open their stomata less often and therefore tin ready well-nigh twice as much net carbon as C3 plants per unit of water lost. Although the vast majority of plant species are C3 plants, C4 plants such equally corn and sugar pikestaff are much more than effective at converting sunlight energy into biomass than C3 plants such as cereal grains. They are therefore of special importance in earth agronomics.
Photosynthesis Depends on the Photochemistry of Chlorophyll Molecules
Having discussed the carbon-fixation reactions, nosotros now return to the question of how the photosynthetic electron-transfer reactions in the chloroplast generate the ATP and the NADPH needed to drive the production of carbohydrates from CO2 and H2O. The required energy is derived from sunlight absorbed by chlorophyll molecules (Figure 14-41). The procedure of energy conversion begins when a chlorophyll molecule is excited by a quantum of light (a photon) and an electron is moved from one molecular orbital to another of college energy. Every bit illustrated in Figure 14-42, such an excited molecule is unstable and tends to return to its original, unexcited state in 1 of 3 ways:
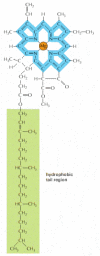
Figure xiv-41
The structure of chlorophyll. A magnesium atom is held in a porphyrin ring, which is related to the porphyrin ring that binds fe in heme (see Effigy fourteen-22). Electrons are delocalized over the bonds shown in blue.

Figure xiv-42
3 ways for an excited chlorophyll molecule to return to its original, unexcited state. The low-cal energy absorbed past an isolated chlorophyll molecule is completely released every bit light and rut by process 1. In photosynthesis, by contrast, chlorophylls (more...)
- 1.
-
By converting the extra energy into heat (molecular motions) or to some combination of rut and low-cal of a longer wavelength (fluorescence), which is what happens when calorie-free energy is absorbed past an isolated chlorophyll molecule in solution.
- 2.
-
By transferring the energy—merely non the electron—direct to a neighboring chlorophyll molecule past a process called resonance energy transfer.
- iii.
-
By transferring the loftier-energy electron to some other nearby molecule, an electron acceptor, and then returning to its original state past taking up a low-energy electron from some other molecule, an electron donor.
The last two mechanisms are exploited in the process of photosynthesis.
A Photosystem Consists of a Reaction Center Plus an Antenna Complex
Multiprotein complexes called photosystems catalyze the conversion of the light energy captured in excited chlorophyll molecules to useful forms. A photosystem consists of two closely linked components: an antenna circuitous, consisting of a big set of pigment molecules that capture light energy and feed information technology to the reaction center; and a photochemical reaction center, consisting of a complex of proteins and chlorophyll molecules that enable lite free energy to be converted into chemic free energy (Effigy 14-43).
Effigy xiv-43
The antenna circuitous and photochemical reaction eye in a photosystem. The antenna circuitous is a collector of lite free energy in the form of excited electrons. The free energy of the excited electrons is funneled, through a series of resonance energy transfers, (more...)
The antenna complex is of import for capturing light. In chloroplasts it consists of a number of distinct membrane protein complexes (known equally light-harvesting complexes); together, these proteins bind several hundred chlorophyll molecules per reaction centre, orienting them precisely in the thylakoid membrane. Depending on the establish, different amounts of accessory pigments called carotenoids, which protect the chlorophylls from oxidation and tin can aid collect lite of other wavelengths, are as well located in each complex. When a chlorophyll molecule in the antenna circuitous is excited, the energy is rapidly transferred from ane molecule to another by resonance free energy transfer until it reaches a special pair of chlorophyll molecules in the photochemical reaction center. Each antenna complex thereby acts as a funnel, collecting low-cal free energy and directing information technology to a specific site where it tin be used finer (see Figure 14-43).
The photochemical reaction center is a transmembrane protein-pigment complex that lies at the heart of photosynthesis. Information technology is thought to have evolved more than 3 billion years ago in primitive photosynthetic bacteria. The special pair of chlorophyll molecules in the reaction center acts as an irreversible trap for excitation quanta considering its excited electron is immediately passed to a chain of electron acceptors that are precisely positioned equally neighbors in the same protein circuitous (Figure 14-44). Past moving the high-energy electron rapidly abroad from the chlorophylls, the photochemical reaction eye transfers it to an environment where it is much more than stable. The electron is thereby suitably positioned for subsequent reactions, which require more time to consummate.

Figure 14-44
The arrangement of the electron carriers in a bacterial photochemical reaction center, equally determined by 10-ray crystallography. The pigment molecules shown are held in the interior of a transmembrane protein and are surrounded by the lipid bilayer of the (more...)
In a Reaction Center, Low-cal Energy Captured by Chlorophyll Creates a Strong Electron Donor from a Weak One
The electron transfers involved in the photochemical reactions merely outlined have been analyzed extensively past rapid spectroscopic methods. An enormous amount of detailed information is available for the photosystem of purple leaner, which is somewhat simpler than the evolutionarily related photosystems in chloroplasts. The reaction centre in this photosystem is a large poly peptide-pigment complex that tin can be solubilized with detergent and purified in agile form. In 1985, its complete 3-dimensional structure was adamant past x-ray crystallography (see Effigy 10-38). This structure, combined with kinetic data, provides the best flick we have of the initial electron-transfer reactions that underlie photosynthesis.
The sequence of electron transfers that take identify in the reaction middle of royal bacteria is shown in Figure 14-45. Every bit outlined previously for the full general example (come across Figure 14-43), calorie-free causes a net electron transfer from a weak electron donor (a molecule with a strong affinity for electrons) to a molecule that is a strong electron donor in its reduced course. The excitation energy in chlorophyll that would usually be released as fluorescence or heat is thereby used instead to create a potent electron donor (a molecule carrying a high-energy electron) where none had been before. In the purple bacterium, the weak electron donor used to fill up the electron-deficient pigsty created by a light-induced charge separation is a cytochrome (come across orange box in Figure 14-45); the strong electron donor produced is a quinone. In the chloroplasts of higher plants, a quinone is similarly produced. However, as we hash out side by side, h2o serves as the initial weak electron donor, which is why oxygen gas is released by photosynthesis in plants.

Figure 14-45
The electron transfers that occur in the photochemical reaction heart of a purple bacterium. A like set of reactions occurs in the evolutionarily related photosystem II in plants. At the top left is an orientating diagram showing the molecules that (more...)
Noncyclic Photophosphorylation Produces Both NADPH and ATP
Photosynthesis in plants and cyanobacteria produces both ATP and NADPH straight past a ii-step procedure chosen noncyclic photophosphorylation. Considering 2 photosystems—called photosystems I and II—are used in series to energize an electron, the electron tin be transferred all the way from water to NADPH. Every bit the high-free energy electrons pass through the coupled photosystems to generate NADPH, some of their energy is siphoned off for ATP synthesis.
The offset of the two photosystems—paradoxically called photosystem II for historical reasons—has the unique ability to withdraw electrons from water. The oxygens of two water molecules bind to a cluster of manganese atoms in a poorly understood water-splitting enzyme. This enzyme enables electrons to be removed one at a fourth dimension from the water, every bit required to fill the electron-deficient holes created by light in chlorophyll molecules in the reaction center. Equally soon as four electrons take been removed from the two water molecules (requiring four quanta of calorie-free), Oii is released. Photosystem II thus catalyzes the reaction 2H2O + 4 photons → 4H+ + 4e - + Otwo. Equally nosotros discussed for the electron-ship chain in mitochondria, which uses Otwo and produces water, the mechanism ensures that no partly oxidized water molecules are released as dangerous, highly reactive oxygen radicals. Essentially all the oxygen in the World's atmosphere has been produced in this way.
The cadre of the reaction center in photosystem Two is homologous to the bacterial reaction center just described, and information technology likewise produces strong electron donors in the form of reduced quinone molecules dissolved in the lipid bilayer of the membrane. The quinones pass their electrons to a H+ pump chosen the cytochrome bhalf dozen-f circuitous, which resembles the cytochrome b-c i circuitous in the respiratory concatenation of mitochondria. The cytochrome b 6 -f complex pumps H+ into the thylakoid space across the thylakoid membrane (or out of the cytosol beyond the plasma membrane in cyanobacteria), and the resulting electrochemical slope drives the synthesis of ATP by an ATP synthase (Figure 14-46).

Figure xiv-46
Electron menses during photosynthesis in the thylakoid membrane. The mobile electron carriers in the concatenation are plastoquinone (which closely resembles the ubiquinone of mitochondria), plastocyanin (a pocket-sized copper-containing protein), and ferredoxin (a small (more...)
The concluding electron acceptor in this electron-transport chain is the second photosystem, photosystem I, which accepts an electron into the electron-deficient hole created by low-cal in the chlorophyll molecule in its reaction eye. Each electron that enters photosystem I is finally boosted to a very high-energy level that allows it to exist passed to the iron-sulfur center in ferredoxin and and then to NADP+ to generate NADPH (Figure xiv-47).

Figure xiv-47
Changes in redox potential during photosynthesis. The redox potential for each molecule is indicated by its position forth the vertical axis. In photosystem II, the excited reaction eye chlorophyll has a redox potential high enough to withdraw electrons (more...)
The scheme for photosynthesis just discussed is known every bit the Z scheme. By means of its two electron-energizing steps, one catalyzed by each photosystem, an electron is passed from water, which normally holds on to its electrons very tightly (redox potential = +820 mV), to NADPH, which usually holds on to its electrons loosely (redox potential = -320 mV). There is not enough energy in a single quantum of visible lite to energize an electron all the way from the bottom of photosystem 2 to the meridian of photosystem I, which is presumably the free energy modify required to pass an electron efficiently from water to NADP+. The use of ii separate photosystems in series ways that the free energy from two quanta of light is available for this purpose. In improver, there is plenty energy left over to enable the electron-transport concatenation that links the two photosystems to pump H+ across the thylakoid membrane (or the plasma membrane of blue-green alga), and so that the ATP synthase can harness some of the light-derived energy for ATP production.
Chloroplasts Tin can Brand ATP by Cyclic Photophosphorylation Without Making NADPH
In the noncyclic photophosphorylation scheme just discussed, high-energy electrons leaving photosystem Ii are harnessed to generate ATP and are passed on to photosystem I to drive the product of NADPH. This produces slightly more than 1 molecule of ATP for every pair of electrons that passes from H2O to NADP+ to generate a molecule of NADPH. Only one.5 molecules of ATP per NADPH are needed for carbon fixation (see Figure xiv-39). To produce extra ATP, the chloroplasts in some species of plants can switch photosystem I into a cyclic mode and so that information technology produces ATP instead of NADPH. In this procedure, called cyclic photophosphorylation, the high-energy electrons from photosystem I are transferred to the cytochrome b 6 -f complex rather than being passed on to NADP+. From the b 6 -f complex, the electrons are passed back to photosystem I at a low energy. The but cyberspace upshot, besides the conversion of some light energy to rut, is that H+ is pumped beyond the thylakoid membrane by the b half-dozen -f complex as electrons laissez passer through it, thereby increasing the electrochemical proton slope that drives the ATP synthase. (This is analogous to the right side of the diagram for purple nonsulfur bacteria in Effigy fourteen-71, beneath.)
To summarize, cyclic photophosphorylation involves just photosystem I, and it produces ATP without the formation of either NADPH or O2. The relative activities of circadian and noncyclic electron flows tin be regulated past the jail cell to determine how much light energy is converted into reducing power (NADPH) and how much into high-energy phosphate bonds (ATP).
Photosystems I and Ii Have Related Structures, and Besides Resemble Bacterial Photosystems
The mechanisms of fundamental cell processes such every bit DNA replication or respiration more often than not turn out to be the same in eucaryotic cells and in bacteria, even though the number of protein components involved is considerably greater in eucaryotes. Eucaryotes evolved from procaryotes, and the additional proteins presumably were selected for during evolution considering they provided an extra degree of efficiency and/or regulation that was useful to the cell.
Photosystems provide a clear example of this type of evolution. Photosystem II, for case, is formed from more 25 different protein subunits, creating a large assembly in the thylakoid membrane with a mass of about 1 million daltons. The atomic structures of the eucaryotic photosystems are being revealed by a combination of electron and 10-ray crystallography. The chore is difficult considering the complexes are big and embedded in the lipid bilayer. Nevertheless, equally illustrated in Figure fourteen-48, the close relationship of photosystem I, photosystem II, and the photochemical reaction heart of purple bacteria has been conspicuously demonstrated from these atomic-level analyses.

Figure 14-48
Iii types of photosynthetic reaction centers compared. Pigments involved in calorie-free harvesting are colored green; those involved in the central photochemical events are colored carmine. (A) The photochemical reaction eye of purple bacteria, whose detailed (more...)
The Proton-Motive Strength Is the Same in Mitochondria and Chloroplasts
The presence of the thylakoid space separates a chloroplast into three rather than the two internal compartments of a mitochondrion. The cyberspace event of H+ translocation in the two organelles is, however, similar. As illustrated in Effigy 14-49, in chloroplasts, H+ is pumped out of the stroma (pH eight) into the thylakoid infinite (pH ~5), creating a gradient of 3–3.5 pH units. This represents a proton-motive strength of about 200 mV beyond the thylakoid membrane, and it drives ATP synthesis by the ATP synthase embedded in this membrane. The forcefulness is the same as that beyond the inner mitochondrial membrane, but virtually all of it is contributed by the pH gradient rather than past a membrane potential, different the instance in mitochondria.

Figure fourteen-49
A comparison of the menstruation of H+ and the orientation of the ATP synthase in mitochondria and chloroplasts. Those compartments with similar pH values have been colored the same. The proton-motive strength across the thylakoid membrane consists almost entirely (more than...)
Similar the stroma, the mitochondrial matrix has a pH of about 8. This is created past pumping H+ out of the mitochondrion into the cytosol (pH ~7) rather than into an interior infinite in the organelle. Thus, the pH gradient is relatively small, and most of the proton-motive strength beyond the inner mitochondrial membrane is instead acquired by the resulting membrane potential (run into Effigy 14-13).
For both mitochondria and chloroplasts, the catalytic site of the ATP synthase is at a pH of about 8 and is located in a large organelle compartment (matrix or stroma) that is packed full of soluble enzymes. Consequently, it is here that all of the organelle's ATP is fabricated (see Figure 14-49).
Carrier Proteins in the Chloroplast Inner Membrane Control Metabolite Exchange with the Cytosol
If chloroplasts are isolated in a fashion that leaves their inner membrane intact, this membrane can be shown to accept a selective permeability, reflecting the presence of specific carrier proteins. Most notably, much of the glyceraldehyde 3-phosphate produced past COii fixation in the chloroplast stroma is transported out of the chloroplast by an efficient antiport system that exchanges iii-carbon sugar phosphates for an inwards flux of inorganic phosphate.
Glyceraldehyde 3-phosphate normally provides the cytosol with an arable source of sugar, which is used past the cell as the starting point for many other biosyntheses—including the production of sucrose for export. Simply this is not all that this molecule provides. Once the glyceraldehyde 3-phosphate reaches the cytosol, it is readily converted (by part of the glycolytic pathway) to 1,3-phosphoglycerate then 3-phosphoglycerate (see p. 97), generating one molecule of ATP and one of NADH. (A similar two-step reaction, but working in contrary, forms glyceraldehyde 3-phosphate in the carbon-fixation wheel; encounter Figure 14-39.) As a result, the consign of glyceraldehyde 3-phosphate from the chloroplast provides not just the principal source of fixed carbon to the residuum of the prison cell, but also the reducing power and ATP needed for metabolism exterior the chloroplast.
Chloroplasts Also Perform Other Crucial Biosyntheses
The chloroplast performs many biosyntheses in improver to photosynthesis. All of the cell'due south fatty acids and a number of amino acids, for case, are made by enzymes in the chloroplast stroma. Similarly, the reducing power of light-activated electrons drives the reduction of nitrite (NO2 -) to ammonia (NH3) in the chloroplast; this ammonia provides the found with nitrogen for the synthesis of amino acids and nucleotides. The metabolic importance of the chloroplast for plants and algae therefore extends far beyond its role in photosynthesis.
Summary
Chloroplasts and photosynthetic leaner obtain high-free energy electrons by means of photosystems that capture the electrons that are excited when sunlight is absorbed by chlorophyll molecules. Photosystems are composed of an antenna complex that funnels energy to a photochemical reaction center, where a precisely ordered complex of proteins and pigments allows the energy of an excited electron in chlorophyll to be captured by electron carriers. The best-understood photochemical reaction center is that of purple photosynthetic bacteria, which comprise just a unmarried photosystem. In contrast, there are two distinct photosystems in chloroplasts and cyanobacteria. The 2 photosystems are normally linked in series, and they transfer electrons from water to NADP+ to form NADPH, with the concomitant product of a transmembrane electrochemical proton gradient. In these linked photosystems, molecular oxygen (O2) is generated equally a by-production of removing 4 low-free energy electrons from two specifically positioned water molecules.
Compared with mitochondria, chloroplasts have an additional internal membrane (the thylakoid membrane) and a third internal space (the thylakoid space). All electron-ship processes occur in the thylakoid membrane: to brand ATP, H+ is pumped into the thylakoid space, and a backflow of H+ through an ATP synthase so produces the ATP in the chloroplast stroma. This ATP is used in conjunction with the NADPH made by photosynthesis to drive a large number of biosynthetic reactions in the chloroplast stroma, including the all-of import carbon-fixation cycle, which creates carbohydrate from CO2. Forth with some other important chloroplast products, this carbohydrate is exported to the cell cytosol, where—as glyceraldehyde 3-phosphate—it provides organic carbon, ATP, and reducing power to the rest of the cell.
Source: https://www.ncbi.nlm.nih.gov/books/NBK26819/
0 Response to "what organelle captures energy from the sun to make food?"
Post a Comment